Tuning Chemistry at the Interface: How Molecular Additives Are Changing CO₂ Reduction
In her PhD at the Interdisciplinary Nanoscience Center and Department of Chemistry, Kirstine Nygaard Kolding explored how small organic molecules can be used to improve copper-based catalysts for CO₂ electroreduction. Her work sheds light on how these additives change the reaction environment and could help make carbon-neutral chemical production more efficient and selective.
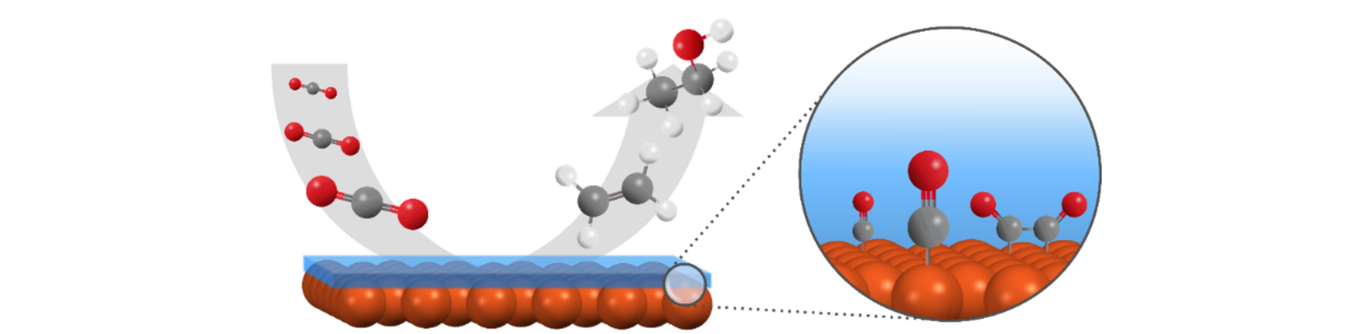
Making Useful Molecules from CO₂
Electrochemical CO₂ reduction is a process that uses electricity to turn carbon dioxide into useful chemicals and fuels. If powered by renewable energy, it could help lower greenhouse gas emissions and support a more sustainable chemical industry. One of the main challenges is designing catalysts that not only drive the reaction efficiently but also help form the products we want, such as ethylene or ethanol, rather than a mixture of other reduction products.
Copper is the only pure metal known to produce larger, multicarbon (C2+) molecules from CO₂. These multicarbon products are of interest as they can be used directly in chemical production or as fuels. But copper alone has limited selectivity. This is where molecular additives offer new possibilities. These are small organic molecules that stay on the surface and change the local environment of the metal surface.
Kirstine’s PhD research focused on understanding how these additives influence the reaction and how they can be used to steer it toward more valuable products.
Key Findings
- Organic additives can help guide the CO₂ conversion process by changing the conditions near the catalyst surface
- Additives may affect not only the reaction but also the catalyst’s structure during preparation
- Real-time measurements reveal the effects of surface additives on the behavior and accumulation of reaction intermediates at the surface during CO2 reduction
- Designing additives that can tailor the environment around active sites (for example, through electronic or secondary coordination sphere effects) is a promising approach to improving catalyst performance
Surface Engineering with Molecular Additives
The concept is to adjust how molecules such as CO2 interact with the catalyst by changing the interface between the metal and the electrolyte. The additives Kirstine studied were mostly organic molecules that interact with the copper surface and/or reaction intermediates.
Her research showed that even very small amounts of these additives can impact the catalyst’s performance. The additives can influence surface structure, shift electronic properties, or stabilize certain intermediates. Each of these effects helps determine the products formed during the reaction.
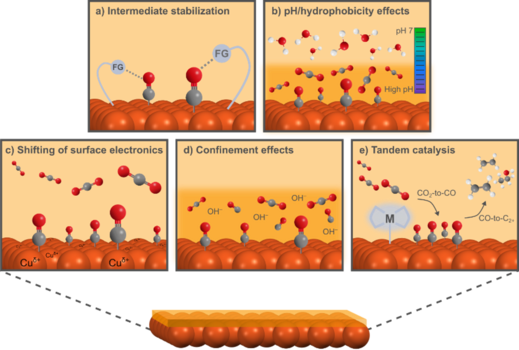
In some cases, additives helped reduce side reactions such as hydrogen evolution. In others, they improved selectivity toward specific multicarbon products. This type of surface tuning makes it possible to optimize CO₂ reduction systems using an affordable and abundant metal as the catalyst.
Case Study: Additive-Controlled Catalyst Activity in Electrochemical CO2 Reduction
In one part of her project, Kirstine examined how hybrid copper catalysts prepared by electrodeposition of organic additives onto copper surfaces performed in the CO2 reduction reaction. She demonstrated that adding specific molecules during the catalyst preparation step affects the nature of the copper surface that forms.
This approach allows control over catalyst properties not just during the reaction but already during preparation. For example, by introducing different oxidation states at the Cu surface, it becomes possible to improve both the selectivity and activity of the catalyst.
How Did They Do It?
To study how additives affect catalytic behavior, Kirstine used in situ Raman spectroscopy to detect important reaction intermediates and an electrochemical voltammetry technique for surface charge determination. These tools allowed her to observe changes in the catalyst interface and reaction intermediates under real operating conditions.
By combining this with electrochemical testing, she could see how different additives shifted product distributions or reaction pathways. Her work suggests that these effects arise from a shift in the electronic state of the copper surface which leads to the stabilization of specific reduction intermediates. For example, the surface-bound CO intermediate which is crucial to formation of the desired multicarbon products.
Why Does It Matter?
Molecular additives offer a simple and flexible method to improve catalyst selectivity and activity without using rare metals or complex nanostructures. Kirstine’s work shows how small changes at the catalyst interface can help guide reactions toward useful products.
This insight is important for building efficient CO₂ conversion systems that are scalable and compatible with real-world conditions. It also contributes to ongoing efforts to close the carbon cycle and support more sustainable chemical production.
If you are working on related materials or applications, we invite you to reach out to our Center Manager to discuss potential collaborations or shared research opportunities.
More information in the following publications:
Kirstine Nygaard Kolding, Kristian Torbensen and Alonso Rosas-Hernández
Kirstine Nygaard Kolding, Jari Leemans, Raffaella Buonsanti and Alonso Rosas-Hernández
Kirstine Nygaard Kolding, Matias Bretlau, Siqi Zhao, Marcel Ceccato, Kristian Torbensen, Kim Daasbjerg, Alonso Rosas-Hernández
More information in the following publications:
Kirstine Nygaard Kolding, Kristian Torbensen and Alonso Rosas-Hernández
Kirstine Nygaard Kolding, Jari Leemans, Raffaella Buonsanti and Alonso Rosas-Hernández
Kirstine Nygaard Kolding, Matias Bretlau, Siqi Zhao, Marcel Ceccato, Kristian Torbensen, Kim Daasbjerg, Alonso Rosas-Hernández
