Small Ions, Big Impact on Gold Nanoparticle Formation
New research from researchers at Aarhus University shows how simple changes in counterions during synthesis affect the stability and quality of gold nanoparticles.
Gold nanoparticles are widely used in sensing, medicine, catalysis, and optics because of their unique physical and chemical properties. Producing them reliably, however, can be challenging. Even well-established synthesis methods sometimes fail unexpectedly or yield inconsistent results. A new study shows that part of this unpredictability may come from an overlooked source: the choice of metal counterions in the reaction.
Researchers at Aarhus University and collaborators found that simply changing the accompanying metal ion in standard gold nanoparticle recipes can have a major effect on nanoparticle quality. In particular, swapping the common sodium or potassium salts for lithium-based salts made the particles more stable, more uniform in size, and more resistant to clumping. These findings provide a new design principle for colloidal synthesis and could help make nanoparticle production more robust, reproducible, and scalable.
Key Findings
- The study provides clear design rules for choosing reagents in gold nanoparticle chemistry
- Gold nanoparticles synthesized with lithium ions are more stable and well-defined than those made with sodium or potassium ions
- The stabilizing effect of lithium was most visible under challenging synthesis conditions (e.g., high precursor concentration).
- The type of cation used affects the growth and stability of nanoparticles across multiple reaction types, including both citrate- and borohydride-based methods.
A Subtle but Powerful Change
In colloidal gold nanoparticle synthesis, the gold salt (usually HAuCl4) is reduced in the presence of stabilizers like citrate or surfactants. These recipes almost always use sodium salts, such as sodium citrate, because they are inexpensive and widely available. Until now, the accompanying sodium ion was assumed to play no significant role in particle formation.
The research team decided to test that assumption by systematically replacing sodium with lithium or potassium in both the citrate-based and borohydride-based synthesis routes. The results were striking. Lithium-based syntheses consistently yielded smaller (around 8 nm) and more uniformly sized spherical nanoparticles that remained stable over time. In contrast potassium-based syntheses were more prone to aggregation and shape changes. Sodium salts gave intermediate results. In other words, lithium allows for more variations while giving the same results, whereas potassium required a very narrow window to avoid failure.
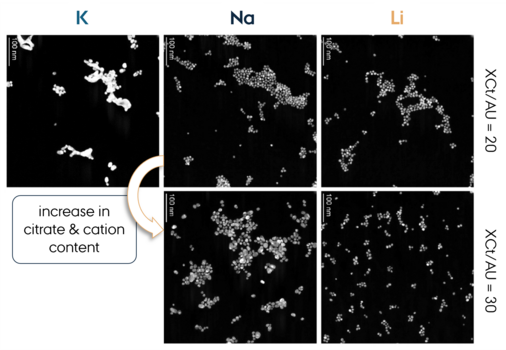
Better Control, Better Science
The lithium-based method did more than improve stability. It also allowed the researchers to push the reaction conditions further. For example, using lithium citrate, they successfully synthesized gold nanoparticles from solutions with five times the usual gold concentration, something that failed when using sodium or potassium citrate. This makes the method more efficient and potentially more scalable, which is especially important for applications requiring large quantities of nanoparticles.
Because the Li⁺-stabilized particles were more uniform and could be made in higher concentrations, they were also better suited for detailed structural analysis. The researchers used small-angle X-ray scattering and total X-ray scattering (PDF analysis) to confirm the internal structure of the nanoparticles. This kind of analysis would not have been possible with less stable particles.
How Did They Do It?
The team combined experiments with a range of analytical techniques:
- UV–Vis spectroscopy tracked how quickly and reliably the characteristic gold nanoparticle color developed in each reaction.
- STEM microscopy was used to measure particle size and shape directly.
- Centrifugation and shelf-life tests revealed which formulations remained stable over time.
- X-ray total scattering and PDF analysis reveal the atomic arrangement and size of particles, using synchrotron radiation.
Why Does It Matter?
This work offers a simple way to improve the reproducibility and scalability of gold nanoparticle production without changing the core chemistry. It also fills a gap in our understanding of colloidal synthesis by showing that counterions, often considered “spectators,” play an active role in stabilizing growing nanoparticles.
By choosing lithium salts instead of sodium or potassium, researchers can produce higher quality nanoparticles and avoid frustrating failures due to hidden recipe sensitivities. This could benefit a wide range of applications that rely on precisely engineered nanomaterials, from drug delivery and diagnostics to plasmonic sensors and catalytic devices.
Read more about their experiments and the detailed characterization in their publication!
You can explore the full study published in Nano Letters.
[1] Positive Thinking: Countercation Effects in Colloidal Syntheses of Gold Nanoparticles
Kristian Junker Andersen, Marton Varga, Aleksandra Smolska, Gregory Nordhal, Jonas H. Jensen, Rodrigo Moreno, Espen D. Bøjesen, Andy S. Anker, and Jonathan Quinson
Read more about their experiments and the detailed characterization in their publication!
You can explore the full study published in Nano Letters.
[1] Positive Thinking: Countercation Effects in Colloidal Syntheses of Gold Nanoparticles
Kristian Junker Andersen, Marton Varga, Aleksandra Smolska, Gregory Nordhal, Jonas H. Jensen, Rodrigo Moreno, Espen D. Bøjesen, Andy S. Anker, and Jonathan Quinson
If you are working on related materials or applications, we invite you to reach out to our Center Manager to discuss potential collaborations or shared research opportunities.
