Layer by Layer: Building Catalysts That Tackle CO₂ and Water Pollution
In her PhD project at Aarhus University, Ronghui Lu set out to design advanced materials that can address two major environmental challenges: pollution in water and excess carbon dioxide in the air. Her research focused on how to use light-driven chemical reactions, known as photocatalysis, to break down organic contaminants and reduce CO₂, using affordable and tunable materials.
Improving LDHs for Environmental Applications
Ronghui Lu developed new multifunctional materials that can simultaneously degrade organic pollutants and convert carbon dioxide using visible light. This dual strategy addresses two critical environmental goals relevant to industries working with wastewater, gas treatment, or green chemistry. To achieve this, Lu worked with a family of materials called layered double hydroxides (LDHs). These are made up of positively charged metal hydroxide layers with charge-balancing anions and water molecules in between. What makes them interesting is that their structure can be adjusted. This includes changing the metal composition, the spacing between the layers, and the type of ions in-between. A simplified illustration of the LDH structure is shown in Figure 1.
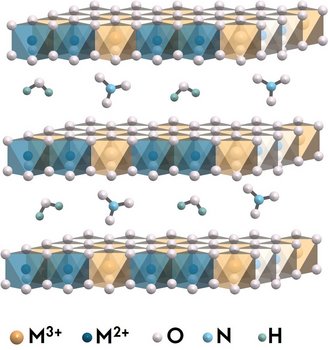
In the first part of her project, she improved the photocatalytic activity of NiFe LDHs for dye removal in water. This was achieved by partially replacing iron cations with bismuth. The resulting Bi-NiFe-LDHs absorbed more visible light and interacted more strongly with pollutant molecules. As a result, the materials not only removed dyes more effectively but also maintained good structural stability under light exposure.
Combining Materials for Dual Functionality
But her goal was broader. She wanted to design a system that could also efficiently reduce CO₂ under light, turning a greenhouse gas into useful products. To achieve this, she combined the modified LDHs with graphitic carbon nitride (g-C₃N₄), a stable and light-absorbing material, to form hybrid systems known as heterostructures. These materials integrate two catalytic functions in one platform using visible light as the only energy source.
Each component contributed its strengths: LDHs provide high surface area, ion exchange capacity, slow recombination rate, and efficient electron flow, while carbon nitride improved visible light absorption and structural stability. Together, they formed heterostructures with a well-connected interface. These interfaces made it easier for electrons and holes to separate, stay apart, and move faster, allowing them to participate in chemical reactions instead of recombining. This significantly boosted the overall photocatalytic efficiency.
Lu then explored how preparation methods, heterostructure composition, and surface treatments influenced the performance of these hybrids. Her adjustments improved the surface area, charge transfer and overall photocatalytic activity. These experiments highlighted the importance of interfacial engineering. A well-connected interface improved electron transport and slowed down unwanted recombination.
Finally, she tested the hybrid catalysts in a closed photocatalytic reactor under visible light. Gas chromatography and NMR analysis confirmed the formation of chemical products such as acetate. The presence of both oxidation and reduction products confirmed the bifunctional activity of the system, where water acts as an electron donor for CO₂ reduction.
How Did They Do It?
To develop and understand these catalysts, Lu used a mix of chemical synthesis, experimental testing, and theoretical modeling:
- Material synthesis by calcination, hydrothermal and exfoliation methods
- UV-Vis, Raman and FT-IR spectroscopy as well as XRD and TEM are used for optical and structural analysis
- Photoluminescence and Photo-Electrochemistry measuring charge separation and transport
- Brunauer–Emmett–Teller theory (BET) and SEM for surface characterization
- Density functional theory (DFT) calculations to explore atomic-scale structure and reaction mechanisms
Key Findings
- Improved dye adsorption and photocatalytic degradation through metal-substitution
- High-performing heterostructures for visible light-driven CO₂ conversion
- Catalyst behavior was tunable through processing conditions such as temperature and ion content.
- Theoretical DFT studies explain how atomic features affected hydrogen activation and electron movement
Why Does It Matter?
Lu’s research shows how a single material system can be designed to carry out more than one important environmental function. Materials that can clean water and reduce greenhouse gases under visible light could support cleaner technologies for both energy and environmental applications.
Her work connects materials chemistry with practical challenges and shows how tuning structure at the atomic and molecular level leads to real performance gains.
What’s Next? Stay Tuned!
The results of Lu's PhD are currently in preparation for publication. Stay tuned for more as the findings are released later this year!
If you are working on related materials or applications, we invite you to reach out to our Center Manager to discuss potential collaborations or shared research opportunities.
