From Vacuum Chambers to Devices: Bridging Surface Science and Industrial Catalysis
Understanding how catalysts work at the atomic level is key to designing better technologies for green hydrogen production. In his PhD at the interdisciplinary Nanoscience Center iNANO, Ramadan Chalil Oglou set out to close that gap. By combining surface studies with real-time measurements on nickel foam electrodes, he showed how fundamental insights from ideal surfaces translate directly to working devices.

Water electrolysis splits water into hydrogen and oxygen using electricity — a crucial process for producing sustainable fuels. However, the oxygen evolution reaction (OER), which occurs at the anode, remains a bottleneck due to sluggish reaction kinetics and high energy requirements.
Nickel-based materials are commonly used in industrial electrolyzers due to their affordability and stability, but their catalytic performance still needs optimization. Understanding how surface chemistry, morphology, and environmental factors influence OER activity is essential to push the limits of efficiency.
Key Findings
Iron often comes from tiny impurities in the electrolyte and gets absorbed into the nickel surface during operation
Nickel surfaces that are rougher and have more edges are better at speeding up reactions and make better use of added iron
Iron only helps the reaction if it ends up in the right places on the nickel surface
Real nickel foam electrodes change in the same way as smooth, lab-grown surfaces when used for water splitting
Why Surface Structure Matters
Catalysts are not just about composition but also about structure. Nickel atoms on flat surfaces behave differently than those at step edges or defects. Ramadan showed that on flat nickel surfaces, iron atoms can actually slow down the activation process, but on stepped surfaces they help form the active NiOOH phase that drives the oxygen evolution reaction. This helped explain conflicting reports in the literature on whether iron helps or hinders nickel-based catalysts. It turns out both can be true, it depends on the structure of the surface underneath.
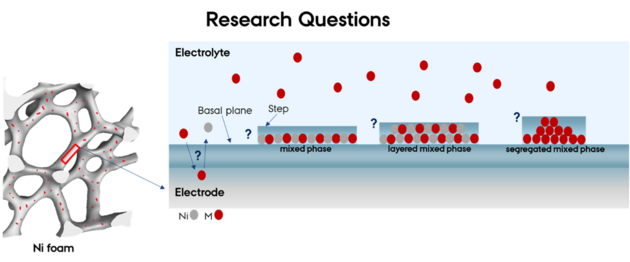
From Model Surfaces to Real Electrodes
Nickel foam is used in commercial electrolysers, but its complex, porous structure makes it difficult to study. To compare real and ideal systems, Ramadan used near-ambient pressure X-ray photoelectron spectroscopy (NAP-XPS) to look at nickel foam electrodes while they were operating in conditions similar to electrolysis.
He found that the nickel foam surface undergoes the same transformation as the model systems. Iron from the electrolyte incorporates into the surface and promotes the formation of layered nickel oxyhydroxides, which are known to be active for oxygen evolution. This confirmed that what happens on clean nickel crystals in a vacuum also happens in more complex and realistic systems.
How Did They Do It?
To understand how nickel catalysts activate and how surface structure and iron affect their behavior, Ramadan used:
- Scanning Tunneling Microscopy (STM) to visualize the structure of nickel single crystals at the atomic scale and identify where iron prefers to bind
- X-ray Photoelectron Spectroscopy (XPS) to determine the chemical state of nickel and iron before and after exposure to electrochemical conditions
- Operando NAP-XPS to monitor real-time changes in the surface of nickel foam under humid, near-working conditions
- Electrochemical testing to measure catalytic performance and link surface chemistry to activity
Why Does It Matter?
Ramadan’s research shows that the structure and chemistry of nickel catalysts change during operation in ways that are predictable from surface science. His work connects model systems to industrial electrodes, helping us understand how to improve catalytic activity and stability in alkaline water electrolysis.
By combining techniques across different length scales and environments, he provided a more complete picture of how nickel catalysts work. These insights can guide the design of more efficient electrodes for hydrogen production and support the transition to clean energy.
If you are working on related materials or applications, we invite you to reach out to our Center Manager to discuss potential collaborations or shared research opportunities.
