Catalyst Combinations That Work: What Real-Time Studies Reveal About Performance and Stability
In his PhD at the Department of Chemistry, Andreas Dueholm Bertelsen explored how combining different metals into complex alloys can improve catalysts for green technologies. His research focused on understanding how these materials form and perform, using real-time measurements to uncover what makes some combinations more stable and effective than others.
Finding the Right Mix
Catalysts are materials that speed up chemical reactions without being consumed. They are used everywhere from industrial manufacturing to car exhaust systems. In green technologies, they play a central role in making hydrogen from water and converting carbon dioxide into fuels or chemicals using electricity.
But catalysts must meet demanding requirements. They need to be efficient, long-lasting, and made from materials that are available at scale. This is especially important in technologies like water electrolysis, where today's best-performing catalysts rely on rare and expensive elements like iridium.
One approach to solving this is to design catalysts made from multiple metals. These materials, sometimes called complex solid solutions or high-entropy alloys, can offer new ways to balance performance and durability. But figuring out which metal combinations actually work is a challenge.
Key Findings
Andreas explored three different types of catalyst materials:
- Multimetal platinum alloys have a noticeable improvement in stability under harsh water-splitting conditions compared to simpler catalysts
- Palladium–copper alloys showed how different metal ratios change the products formed from CO₂
- Iridium–ruthenium oxides delivered high performance while reducing use of the rare metal iridium
The results suggest that using multiple metals in a single material can not only boost performance but also improve stability, which is essential for industrial applications.
Three Ways Metal Combinations Improve Catalyst Performance
Below, we take a closer look at what the real-time experiments revealed about each type of catalyst. To understand why some metal combinations form better than others, Andreas used real-time X-ray techniques that allowed him to study the catalyst formation. These measurements helped reveal what is often impossible to see with conventional methods.
How Mixing Several Metals Creates Stronger Hydrogen Catalysts
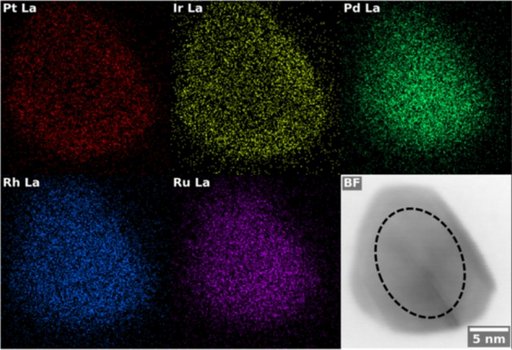
For the platinum-based multimetal alloys, the real-time studies showed that platinum plays a key role in helping many elements mix into a single, uniform structure. The result is a stable alloy that withstands the harsh conditions inside a water electrolyser. He worked on multi-metal nanoparticles that contained up to five platinum-group metals: Platinum (Pt), Iridium (Ir), Palladium (Pd), Rhodium (Rh) and Ruthenium (Ru). Adding more types of metals did weaken activity of the catalyst slightly, but, in fact, the five-metal combination turned out to be much more durable than simpler three or four-metal alloys. This shows that complexity, when carefully designed, can potentially make catalysts more robust.
Efficient Oxide Catalysts That Use Less Rare Material
For the iridium and ruthenium oxides, the real-time measurements helped uncover how these materials form at the atomic level. Mixing iridium with ruthenium created tiny oxide particles that combined the efficiency of ruthenium with the stability of iridium. This means it is possible to use less of the rare iridium while still keeping performance high, which is valuable for scaling hydrogen production.
Metal Ratios That Change the Pathway of CO₂ Conversion
In the copper and palladium system, Andreas found that even small changes in composition made a big difference in what the catalyst produced. Copper is known for turning CO₂ into valuable hydrocarbons, while palladium is better at producing simpler molecules. By adjusting how much palladium was added, he showed how the reaction pathway could be steered in different directions. This is useful when industries want to control exactly which products are formed. This means the catalyst can be tailored for different industrial goals, such as producing fuels, chemicals, or feedstocks.
Together, these results show how real-time experimentation can guide researchers toward more robust and efficient catalyst designs. They also highlight how multi-metal combinations create opportunities that single-element materials cannot achieve.
How Did They Do It?
To understand why different metal combinations, behave the way they do, Andreas combined real time X-ray techniques with laboratory synthesis and electrochemical testing:
- In situ X-ray diffraction and total scattering (PDF) were used to watch the materials form during synthesis. These measurements showed when metals mixed into one phase and how structure influenced stability and activity.
- He used different approaches for controlled nanoparticle synthesis making it possible to compare how different preparation routes affected particle size, mixing, and overall quality.
- Electron microscopy and elemental mapping (STEM-EDX) helped confirm that the metals were evenly distributed inside each nanoparticle.
- Electrochemical testing allowed Andreas to evaluate how well the catalysts performed in key reactions. This included oxygen evolution for hydrogen production and CO₂ reduction for producing valuable chemicals.
Together, these tools provided a complete picture: how the catalysts form, how their structure develops, and how those structures translate into performance in clean energy technologies.
Why Does It Matter?
As countries and industries look to reduce carbon emissions, new materials are needed to support clean energy technologies. Andreas' research helps bridge the gap between fundamental chemistry and practical applications. By showing which metal combinations are stable, efficient, and easier to produce, the findings offer a roadmap for designing better catalysts for future energy systems.
His work highlights the value of real-time experiments and comparative studies. These approaches not only uncover what works at the atomic level but also guide the development of scalable and durable solutions that industry can rely on.
If you are working on related materials or applications, we invite you to reach out to our Center Manager to discuss potential collaborations or shared research opportunities.
More information in the following publications:
Andreas Dueholm Bertelsen, Magnus Kløve, Nils Lau Nyborg Broge, Martin Bondesgaard, Rasmus Baden Stubkjær, Ann-Christin Dippel, Qinyu Li, Richard Tilley, Mads Ry Vogel Jørgensen, Bo Brummerstedt Iversen
[2] Copper-rich PdxCu1-x alloy nanoparticles as catalyst for electrochemical reduction of CO2
Andreas Dueholm Bertelsen, Rebekka Klemmt, Kirstine Nygaard Kolding, Espen Drath Bøjesen, Bo Brummerstedt Iversen, Chem. Mater. 2025, 37, 17, 6619–6628
Raul Castellano Perdomo, Andreas Dueholm Bertelsen, Nikolaos Antonios Iakynthos Nemet, Bo Brummerstedt Iversen. This content is a preprint and has not been peer-reviewed.
Andreas Dueholm Bertelsen, Alexander Reinhardt Hansen, Nils Lau Nyborg Broge, Aref Mamakhel, Martin Bondesgaard, Bo Brummerstedt Iversen
[5] Facile Solvothermal Synthesis of Pt–Ir–Pd–Rh–Ru–Cu–Ni–Co High-Entropy Alloy Nanoparticles
Nils Lau Nyborg Broge, Andreas Dueholm Bertelsen, Frederik Søndergaard-Pedersen, Bo Brummerstedt Iversen
